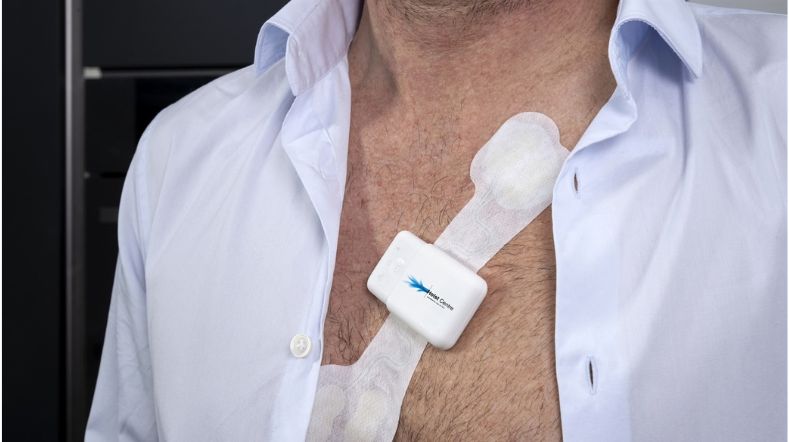
Digital Biomarker Lab: trusted digital biomarker development and validation from daily-life data
The TNO Digital Biomarker Lab reveals meaningful health patterns from daily life through patient-centric digital biomarkers. We turn digital measures into trusted evidence for clinical research and healthcare decision-making.
Why digital biomarkers
Across everyday life, care pathways and clinical studies, people generate rich signals about health, behaviour and treatment response. Yet these signals are rarely captured, validated or translated into meaningful evidence that patients, clinicians and decision-makers can rely on.
Traditional clinical measures are episodic and often burdensome, capturing only a limited snapshot of a person’s health. Important information about how people function, respond to treatment and recover between visits can be missed, despite its relevance for care and evaluation.
Digital biomarkers offer a promising solution. By collecting objective signals more continuously in daily life, they can complement traditional measures and provide a richer
About Digital Biomarkers
They are also meaningful to patients because they remain grounded in real life. Instead of relying only on abstract questionnaires, digital biomarkers can provide timely, objective and personalised insights into an individual’s activity profile and functioning. When embedded into self-management tools and care pathway structures, these measures can help bridge the gap between patients and healthcare, supporting more informed conversations and decisions.
However, the full potential remains untapped. Many digital measures are not yet supported by the structured evidence needed for adoption, and development efforts are often fragmented across measurement design, validation, usability and data infrastructure. This slows progress from promising signals to tools that are scalable and ready for real-world use, including in regulated clinical development.
From digital measures to validated digital biomarkers
At the TNO Digital Biomarker Lab, we use a structured pathway to develop digital biomarkers that are trustworthy, interpretable, and meaningful in daily life, so they can support decisions in clinical research and healthcare.
We take a rigorous, independent, and patient-centric approach. We start with the question that matters most: what do patients and clinicians need to understand, monitor, or improve? From there, we define the intended context of use and build the evidence package required for adoption, scientifically, operationally, and, where relevant, regulatory-aligned.
Because digital biomarkers are increasingly used in settings where quality and transparency expectations are high, we are regulator-aware in how we design evidence generation. In many settings, digital biomarkers will only be useful at scale if they meet expectations for quality, validation and transparency. We help clients and partners navigate these requirements in practice, so digital biomarkers can move from promising signals to credible, usable tools in the real world.
Building on long-standing experience in measuring health and behaviour, TNO combines scientific depth with practical expertise in digital health and clinical research.
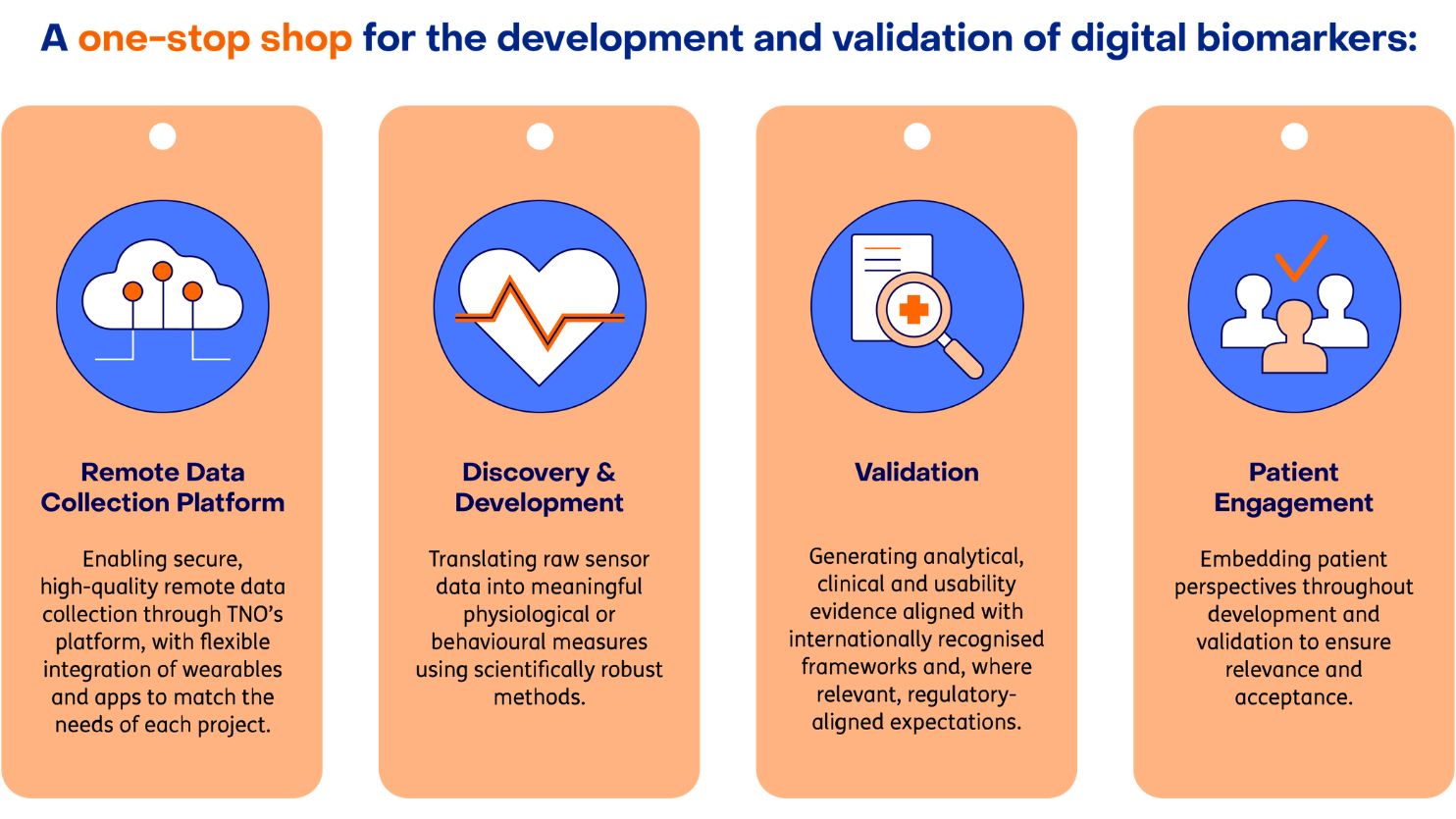
Services: digital biomarker discovery, validation and remote data collection
We support partners across key stages of the digital biomarker lifecycle. By combining discovery, validation, patient engagement and data infrastructure, we enable clients and partners to build digital biomarkers that are robust, interpretable and ready to be used, whether in clinical research, healthcare practice or real-world settings.
Portfolio: turning evidence into impact through projects and partnerships
The TNO Digital Biomarker Lab builds on more than 20 years of expertise in measuring health and behaviour, combined with the team’s focused experience in digital biomarker development across multiple therapeutic areas.
Our projects span chronic inflammatory, cardiovascular, metabolic, neurological and sleep-related conditions, delivered in close collaboration with partners from industry, academia and healthcare. Through scientific rigour and fit-for-purpose digital measures, we translate complex sensor-derived data into meaningful evidence that supports innovation in medicine.
Let’s work together to accelerate digital health innovation
We collaborate with pharmaceutical, MedTech and healthcare partners to advance digital biomarker development, from early discovery to validation and deployment, through various modes:
- Joint research and development projects
- Industry partnerships and service contracts
- Clinical validation and pilot studies
- Strategic advisory and training
Ready to explore what digital biomarkers can do for your project or care pathway? Contact our team to discuss the right evidence pathway and collaboration model.
Get inspired
Beyond the breath count: patients want meaningful digital metrics in sleep apnoea monitoring


DIMPLAD: Empowering early recognition of Alzheimer’s with user-centered digital solutions


IBDigital Project Starts at ECCO Conference in Berlin


How can AI help reduce pressure on our healthcare system?


Health applications