Digital Biomarker Services: Turn sensor data into trusted clinical evidence
As an independent partner, the TNO Digital Biomarker Lab supports partners across the early lifecycle of digital biomarker development, from identifying meaningful measurement opportunities with patients to generating robust, fit-for-purpose evidence for research, clinical studies, and real-world use. We combine scientific rigour with practical implementation expertise to help pharma, MedTech, and research partners reduce uncertainty, make informed decisions, and accelerate progress with confidence.
The challenge: bridging the gap between digital data and clinical evidence
Traditional clinical measures capture only snapshots of health, missing the rich, continuous data that reflects how patients truly respond to treatment in daily life. Digital biomarkers can fill this gap, but without rigorous validation, they remain untrusted and unused.
Regulatory bodies increasingly require patient-centric, digitally derived endpoints. Yet many organisations struggle to navigate the complex path from raw sensor signals to evidence that regulators, clinicians, and payers will accept.
Digital biomarker development: from data to decision
The TNO Digital Biomarker Lab delivers end-to-end support across four interconnected service areas. Each can be deployed independently or combined for comprehensive digital biomarker development.
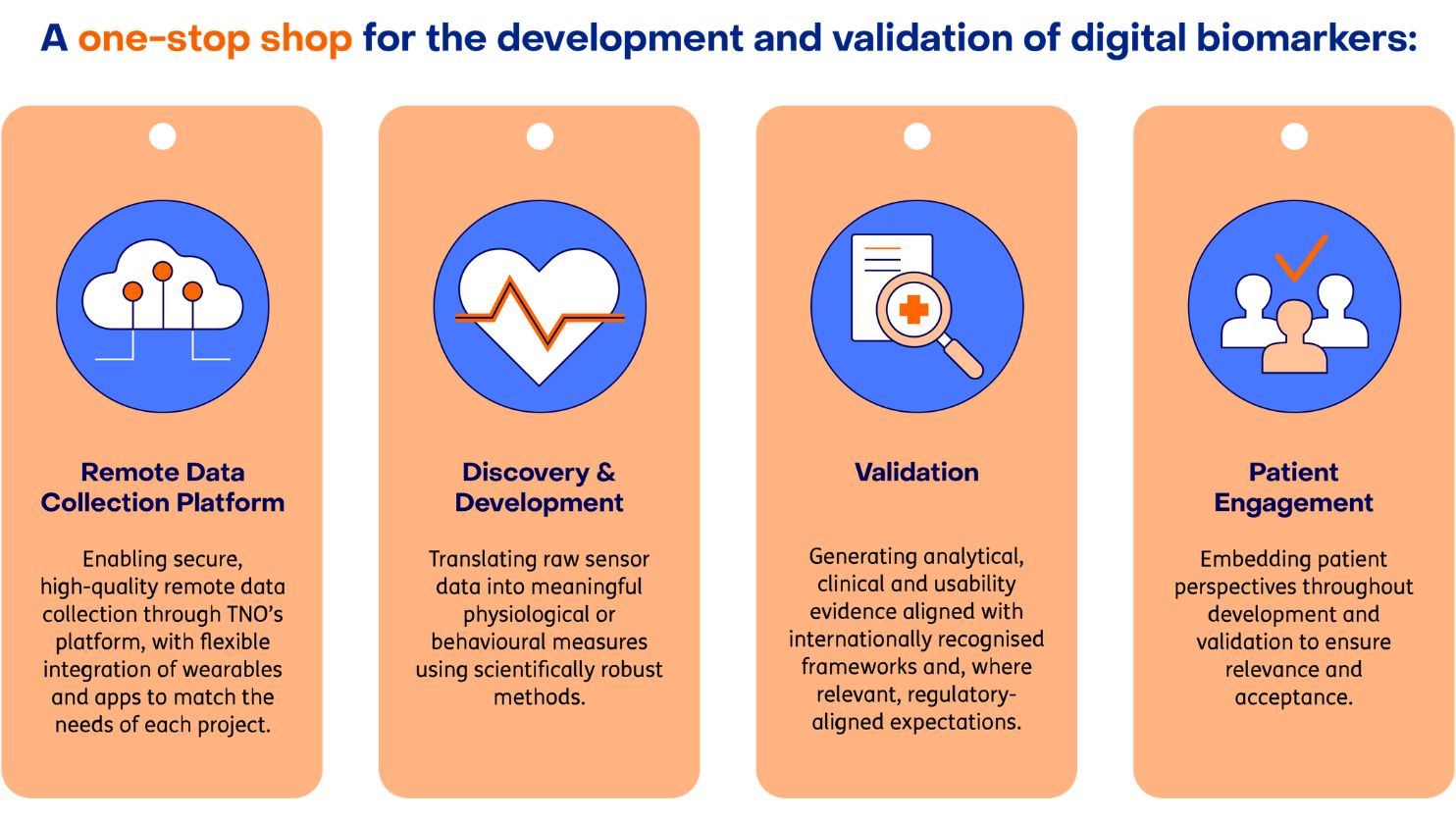
Remote data & collection platform
Reliable digital biomarkers require reliable data collection. TNO’s Remote Data Collection Platform is ISO- and NEN-certified infrastructure for collecting, managing, and integrating data from wearables, smartphone sensors, and questionnaires in clinical and research settings.
The platform’s device-agnostic design gives you the flexibility to select and combine wearables and questionnaires that best fit your specific project needs. Whether running feasibility pilots or large-scale Bring-Your-Own-Device studies, you choose the right sensors and assessments without vendor constraints.
The platform provides full data transparency, giving you complete visibility into data collection, quality metrics, and participant compliance. Real-time dashboards enable continuous monitoring of data integrity throughout the study lifecycle and enhance participant engagement through real-time feedback.
Fragmented digital health ecosystems slow research progress. Healthcare and research organisations lack certified infrastructure that provides both the security needed for sensitive health data and the flexibility required for rigorous scientific studies.
We eliminate infrastructure barriers with device-agnostic, certified infrastructure that enables flexible sensor selection, secure data collection, real-time quality monitoring, and full privacy compliance. You gain the freedom to design optimal studies without building proprietary systems or accepting vendor lock-in.
- Freedom to select and combine the most fit-for-purpose sensors and questionnaires for your project.
- Complete data transparency with real-time quality monitoring and compliance dashboards.
- Study support with participant application and study dashboard.
- Integrated study and participant portals with remote onboarding capabilities.
- ISO27001, NEN7510, and ISO13485 certification for sensitive health data.
Discovery & development
Developing digital biomarkers requires more than algorithms. It requires physiological interpretation, analytical validation, and transparent methods to translate sensor data, like ECG, respiration, activity or continuous glucose into reproducible and clinically relevant measures.
We transform high-frequency wearable sensor data into clinically interpretable digital biomarkers. This includes analytical validation of sensor outputs, signal quality control, advanced signal processing, and context-aware modeling that incorporates physiological dynamics.
Many digital health initiatives generate large volumes of data without the analytical validation and physiological grounding required for clinical adoption. Algorithms remain black boxes, limiting interpretability, regulatory acceptance and clinical trust.
We generate the technical and scientific evidence required to support feasibility assessments and informed go/no-go decisions in early development. Our models are transparent and grounded in physiology, and are tested against established clinical reference standards to demonstrate reliability and real-world relevance.
- Physiology-informed digital biomarkers rather than black-box AI outputs.
- Comprehensive data quality assessment from the start.
- Validated signal processing with quantified agreement to clinical reference standards.
- Transparent, reproducible algorithms suitable for regulatory and scientific scrutiny.
- Early-stage feasibility assessment to reduce development risk.
- Independent, vendor-neutral evaluation.
Validation
Robust evidence is essential for digital biomarkers to be trusted, adopted, and used in healthcare and research. The Digital Biomarker Lab designs and conducts clinical and validation studies in collaboration with clinical partners to assess if digital measures perform reliably and meaningfully in their intended context of use.
We support partners across analytical validation, clinical validation, and usability evaluation, applying internationally recognised frameworks. Our team combines expertise in protocol development, sensor evaluation, data analysis, regulatory strategy, and iterative monitoring, including remote patient monitoring and decentralised trial designs.
By identifying technical, clinical, and usability challenges early, our approach reduces risk, supports regulatory readiness, and generates reliable evidence to inform implementation and scaling.
Many digital biomarkers fail to reach clinical use because they lack robust validation evidence. Regulators, clinicians, and payers require proof that digital measures perform reliably in real-world settings before adoption.
We design and execute analytical, clinical, and usability validation studies with clinical partners, combining protocol development, sensor evaluation, regulatory strategy, and remote monitoring expertise. Our independent position ensures credible, publication-grade evidence.
- Validation studies aligned with the V3+ framework from DiMe and other regulatory frameworks.
- Protocol development.
- Regulatory strategy consultation.
- Support with evidence packages needed for submissions and publications.
Patient engagement
Patient engagement begins from the very start of digital biomarker development. We involve patients early to identify which digital biomarkers are truly meaningful to them, ensuring that development efforts focus on outcomes that matter most in their daily lives. This early involvement shapes study design, endpoint selection, and measurement approaches before significant resources are committed.
Digital biomarkers only succeed when they reflect what matters to patients and are usable in daily life. We integrate patient perspectives from concept through validation, ensuring measures are relevant, acceptable, and aligned with patient priorities.
Digital health technologies often fail because they don’t address patient needs or are too burdensome to use consistently.
Through longstanding partnerships with patient organisations across disease areas, we bring structured patient engagement to every phase of digital biomarker development. Using recognised international frameworks such as the Digital Measures That Matter framework, we design co-creation sessions, advisory boards, and usability assessments. Our in-house social science capabilities ensure methodological rigour throughout.
- Patient input embedded from the earliest stages of biomarker identification and sustained throughout the full development and validation trajectory.
- Focus on outcomes that are meaningful to patients in their daily lives.
- Structured engagement aligned with scientific and regulatory requirements.
- Independent facilitation between industry, clinicians, and patient communities.
- Enhanced adoption and real-world performance.
How to work with us
Flexible engagement models tailored to your needs
1.
Tailored expertise, exactly when you need it: Our single-service engagements give you direct access to specialist knowledge at critical moments. Whether you require support with platform infrastructure, algorithm development, validation studies or patient engagement, we can step in with focused, high-impact expertise.
2.
Integrated programmes for end-to-end success: When your challenge demands more, we bring multiple services together in fully integrated programmes. From discovery through development to validation, our joined-up approach delivers continuity, efficiency and robust scientific progression at every stage.
3.
Strategic advisory that shapes better decisions: Strategy is embedded in everything we do. We work alongside you to navigate unmet needs, refine study design, select the right methodologies and meet evidence requirements - ensuring confident, well-informed decisions throughout your journey.
4.
Let’s start the conversation: Our initial discussions are collaborative and exploratory, designed to understand your context and define the right scope and approach. Get in touch to explore how we can support your ambitions and turn complex challenges into clear, actionable progress.
‘Digital biomarkers only create real impact when they are scientifically validated and clinically meaningful. Translating continuous behavioural data into trusted evidence requires independent expertise and methodological rigour. TNO provides the validation framework and scientific guidance that turn digital innovation into credible, stakeholder-ready solutions.’
Ready to advance your digital biomarker programme?
Whether you’re exploring feasibility, seeking validation evidence, or scaling deployment, the TNO Digital Biomarker Lab provides the independent expertise and certified infrastructure you need. Partner with us to develop, validate, and deploy fit-for-purpose digital biomarkers that regulators, clinicians, and patients trust.