TOMCA: tissue-optical analysis of wearable medical sensors
More and more wearable sensors are being developed that use light to measure health parameters in the skin. The challenge here is to make them sufficiently accurate and reliable for the intended application. When these sensors are used as medical devices it is particularly important that they meet the strict quality requirements. With its extensive knowledge and many years of experience in the field of tissue optics, TNO supports its partners with the design and optimisation of wearable optical sensors. We work together with companies that develop smart watches, health patches or other wearables.

The field of Medical Photonics is developing rapidly. Sensor hardware is becoming increasingly sophisticated and compact, and we are now able to come up with improved, more complex algorithms based on advanced data models.
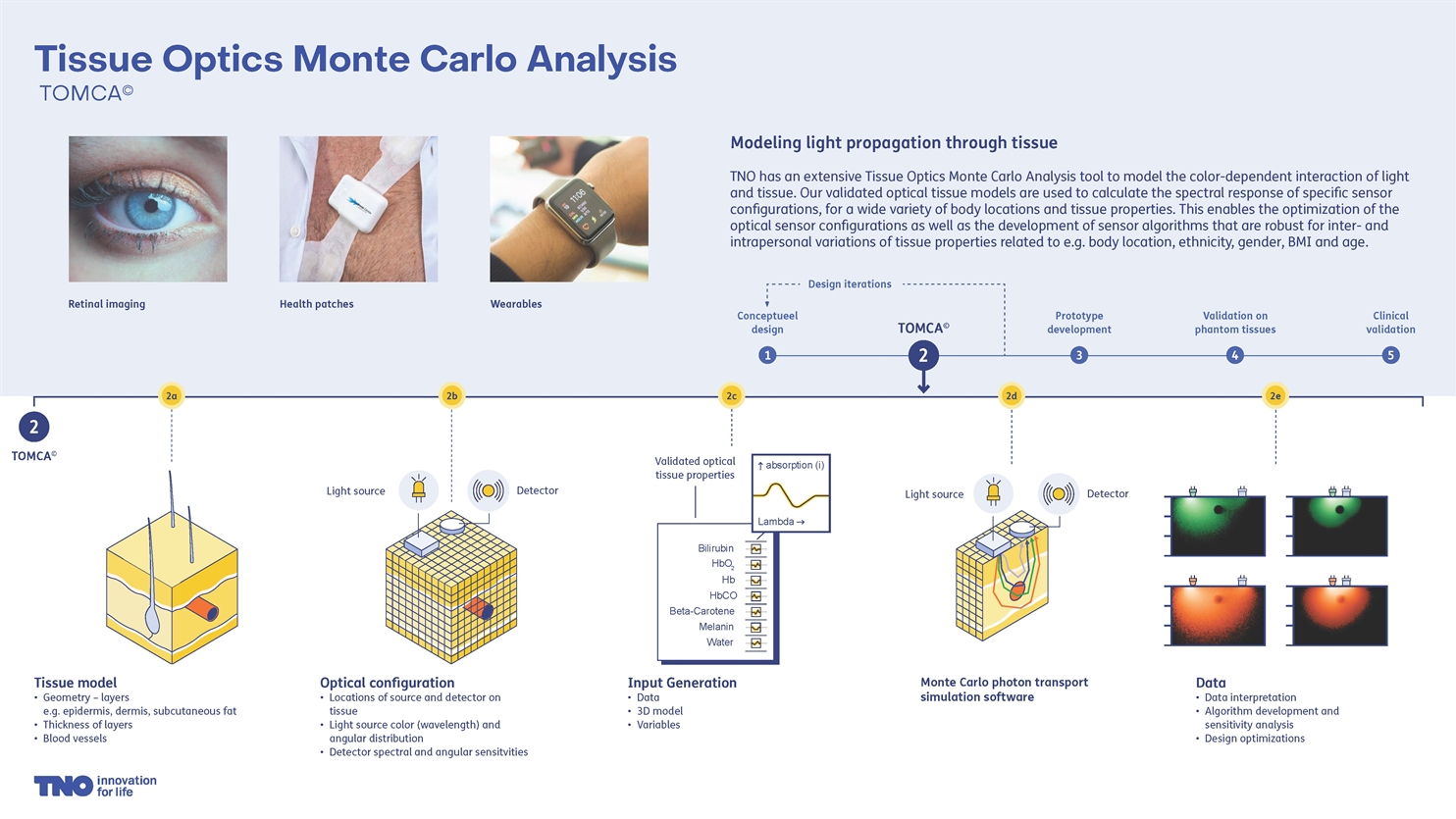
To optimise the physical design and software algorithms of wearable optical sensors, TNO has developed a process that allows the performance of different sensor designs to be analysed on a large number of virtual skin models.
Tissue Optics Monte Carlo Analysis (TOMCA®)
Ensuring both the accuracy and robustness of your sensor algorithm is crucial. The challenge here is that such an algorithm has to be able to take a huge number of human variables into account. After all, everyone has a different skin structure and many skin variables change over time within each skin. Your skin becomes browner if you have been in the sun more, for example, and its blood flow changes depending on the temperature.
These different skin types and their varying composition can strongly influence the accuracy of the health parameter to be measured. A smarter sensor design and algorithm will increase the sensors’ accuracy and suppress disturbing factors more effectively. TNO has an optical Monte-Carlo-based analysis tool: TOMCA®. Using optical models of the skin and the spectra of the substances found in it, it is possible to analyse how sensor configurations interact with the skin at different colours of light.
Voxels
The skin models are composed of different layers and can also incorporate blood vessels. By varying the size of blood vessels, it is possible to simulate pulsations. For each skin model the skin layers are divided into small blocks (voxels). The local optical tissue properties, such as scattering and absorption properties, are defined for each voxel, as are the position and properties of the light source(s) and detector(s).
In this way, using such a 3D skin model makes it possible to predict how the light will be distributed or absorbed within the skin, and therefore also how much light from a source will arrive at a detector and what path this light has probably taken through the skin layers. To allow even better modelling of structures such as blood vessels, TNO is currently moving towards the use of voxels with inclined faces. Such voxels can approximate the curves of blood vessels better and therefore provide a more accurate impression of reality.
50,000 different (simulated) skin types
To develop sensor algorithms, a huge amount of optical response data is needed from a sensor configuration on thousands of different types and conditions of skin. Gathering this information by performing physical measurements on test subjects is far too expensive and time-consuming. That is why TNO performs TOMCA® analyses on thousands of different skin models and conditions. This involves setting a number of supercomputers to work in parallel. When the data is available it may be used to analyse existing algorithms or develop new algorithms. Optimising both the sensor configuration and the sensor algorithm makes the sensor more accurate and robust.
Once the sensor design has been optimised on the skin models using the TOMCA® tool, it is time to validate it in practice. A sensor that has been built is often tested first on phantom tissues, because the optical properties of these are known exactly. After that, clinical validation on human subjects begins.
Strong scientific basis, applied in practice
In the field of Medical Photonics, TNO colleague Prof. Arjen Amelink works together closely with Vrije Universiteit Amsterdam (Prof. Johannes de Boer). TNO is focusing in particular on measuring biomarkers in the skin or retina, such as oxygen saturation (SpO2), haemoglobin, heart rate, blood pressure, beta-carotene, fat content and hydration. In projects TNO often works closely with the R&D departments of industrial partners.
Access to a hypobaric chamber
There are several ways to check the accuracy of the measurement results. For example, TNO works together with the Centre for Man and Aviation (CML) in Soesterberg. There they have a hypobaric chamber that simulates the air pressure at high altitude. This is an ideal environment to check the accuracy of SpO2 sensors in the 100-70% saturation range.
Working on a new generation of medical devices
In addition to improving wearable sensors for clients, TNO also develops new, innovative sensors for wearable devices on its own initiative. We are working, for example, on flexible skin patches: a plaster with a number of light sources, light sensors and a communication antenna to allow the sensor data to be received using a mobile phone. TNO is looking for market parties who are interested in co-investing in such innovations to develop them further into products.
À la carte
TNO manages the process from initial idea through to validation of clinical prototypes: drawing up the requirements for the intended application, design, analysis, realisation and support during clinical validation. Our mission is to support medical parties with the development of their sensors. You can therefore also approach TNO for support for individual process steps on an ‘à la carte’ basis.
Flyer TOMCA®
Knowledge about light-tissue interaction is crucial when designing optical sensors in wearables, smartwatches and retinal cameras. With TOMCA®, TNO simulates how light passes through the tissue and uses this to develop inclusive, robust algorithms.
Get inspired
Optics


Art and quantum: student team renders superposition tangible with TNO


TNO expands quantum information technology testing capabilities to support startups


Sharper choices needed in the Dutch chip industry
TU Delft appoints TNO’s Richard Versluis as Quantum and Computer Engineering Fellow


